Ion Charge: What is it?
An Ion Is A Electrically Charged Atom That Can Either Lose Or Gain An Electron. The Way Ions Form If A Neutral Atom Loses One Or More Electrons, It Has A Net Positive Charge And Is Known As A Cation. If An Atom Gains Electrons, It Has A Net Negative Charge And Is Known As An Cation.
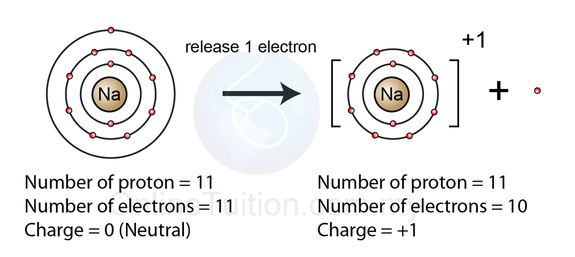
Consider a Sodium atom : A sodium atom has 11 electron and 11 protons but this Sodium atom in many cases can lose an electron to become a Sodium ION. this Sodium ion still has the same number of protons but fewer electrons. Any positively charged ions are called CATIONS.
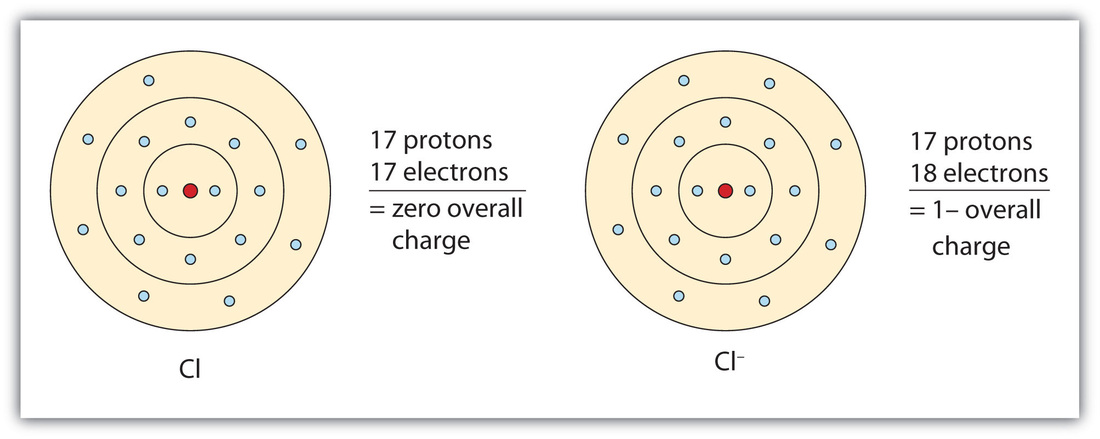
Now let us look at this Chlorine atom: A Chlorine atom has 17 protons and 17 electrons. However a chlorine atom can GAIN an electron to form chlorine ion. the Chloride atom still has the same number protons but has an extra electron. Negatively charged ions are called ANIONS.
ION CHARGE TRENDS
Trend in Ion Charge:
- Alkali Metals always have a +1 Ion Charge
- Alkaline Earth Metals all have a +2 Ion Charge
- Nitrogen and Phosphorus both have a -3 Ion Charge
- Halogens all have -1 Ion charge
- Noble Gases do not an Ion Charge because their valence shells are already full