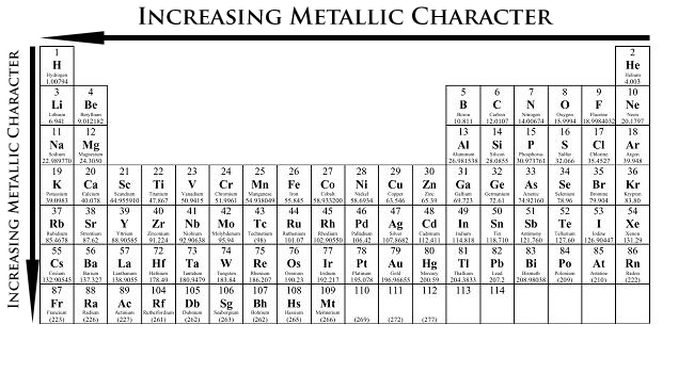
Metallic Character Trends
Metallic Character of an element is how readily an atom can lose an electron. Metallic Character also includes the ability to form ionic chlorides and basic oxides.
Therefore we can say that:
This happens because as you go from left to right of the Periodic Table the attraction between the valence electrons and its nucleus is much weaker, therefore making it easier for the atom to lose electrons. And as you go down a group the bigger atomic radius an element has the weaker its attraction is because its outer shells are much farther away, therefore weakening its ability to keep the electrons.
Therefore we can say that:
- Metallic Character increases from left to right of the Periodic Table
- Metallic Character increases as it goes down the Periodic Table
This happens because as you go from left to right of the Periodic Table the attraction between the valence electrons and its nucleus is much weaker, therefore making it easier for the atom to lose electrons. And as you go down a group the bigger atomic radius an element has the weaker its attraction is because its outer shells are much farther away, therefore weakening its ability to keep the electrons.