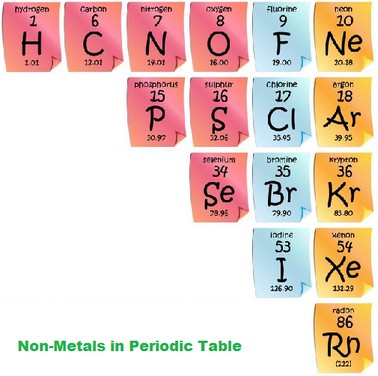
Non-Metals
Non-metals tend to have high ionization energies and electronegativities. Generally speaking they are poor conductors of heat and electricity because there are no metals present in non-metals, and in order to produce heat or electricity you need to heat a metal. therefore in the absence of a metal, non-metals cannot self-heat. Most non metals have the ability to gain electrons easily. Non-metals have relatively low melting and boiling points, with a few exceptions. Non-metals are also very brittle, due to the fact that most non-metals take the form of gases or powders.