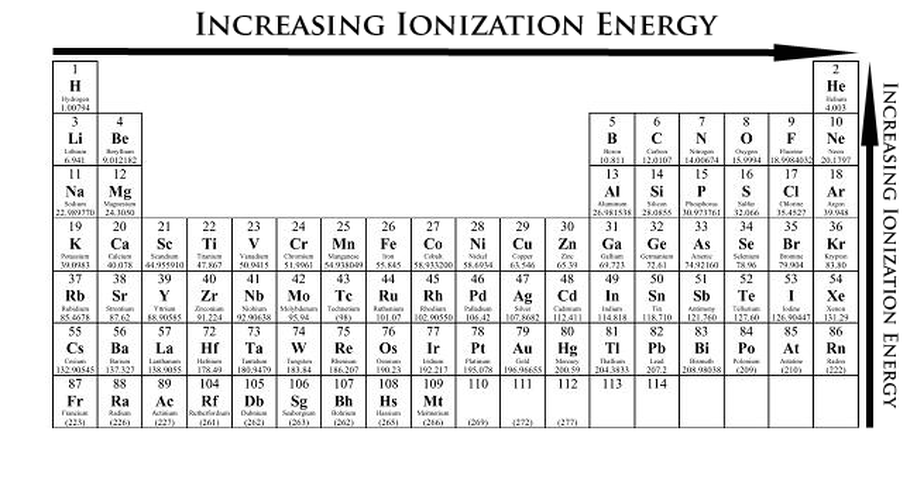
Ionization Energy Trends
Ionization energy is the energy required to remove an electron from a neutral atom in its gaseous phase. Alkali Metals which has one electron on their valence shell, requires less energy from elements who has more than one electrons on their valence shell. Also Elements on the right would rather take electrons rather than to give them up, taking electrons requires much more energy than giving up electrons.
Therefore we can say that:
Shielding affect
The reason as to why Ionization Energy decreases going down the periodic table is because of shielding effect. Shielding Effect is when electron and nucleus in an atom have an decrease in attraction. The shielding affect makes it easier to remove the outer most electrons from those atoms that have many electrons (those near the bottom of the chart). Because as we go down the period there is an increase of electrons it is harder to attract it, there fore the electrons are less attracted to the nucleus, decreasing the Ionization Energy
Finding Ionization Energy:
Some elements have several ionization energies, to calculated their "first ionization energy" we use the equation:
Na --> Na+ + e-
For the second ionization Energy we use:
Na+ --> Na2+ + e-
Therefore we can say that:
- Ionization Energy increases as you left to right of the periodic table
- Ionization Energy decreases as you go down the group
Shielding affect
The reason as to why Ionization Energy decreases going down the periodic table is because of shielding effect. Shielding Effect is when electron and nucleus in an atom have an decrease in attraction. The shielding affect makes it easier to remove the outer most electrons from those atoms that have many electrons (those near the bottom of the chart). Because as we go down the period there is an increase of electrons it is harder to attract it, there fore the electrons are less attracted to the nucleus, decreasing the Ionization Energy
Finding Ionization Energy:
Some elements have several ionization energies, to calculated their "first ionization energy" we use the equation:
Na --> Na+ + e-
For the second ionization Energy we use:
Na+ --> Na2+ + e-