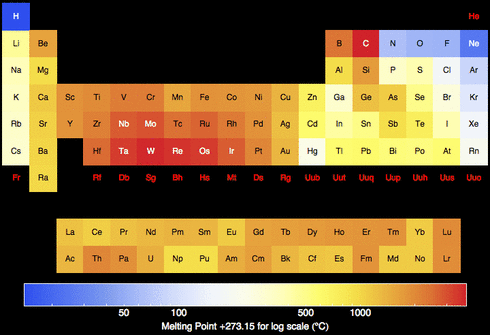
Meltin' Points
A melting point is the amount of energy required to break a bond to change a solid phase of a substance to a liquid.
Obviously the stronger the bond is the more energy it requires to break that bond. Melting points varies and they don't have a trend in the periodic table.
- Metals generally possess a high melting point.
- Most non-metals possess low melting points.
Melting disrupts the arrangements of the atoms, therefore the heat required to break apart the atoms depends on the strength of the attraction. High attraction = More Heat.